Today, Professor Inga Prokopenko tells us about his research projects within PreciDIAB and Mendelian randomization that could change the world of human genetics research.
Within PreciDIAB, I co-lead, together with Amélie Bonnefond and Philippe Froguel, the work package 2.2 on Markers of diabetes treatment failure and of cancer risk. Within this work package, two PhD students Jared Gichohi Maina and Vincent Pascat, are working together with us on advancement of analyses dissecting the shared genetics between metabolic phenotypes and several cancers, including pancreatic, colorectal, breast and prostate.
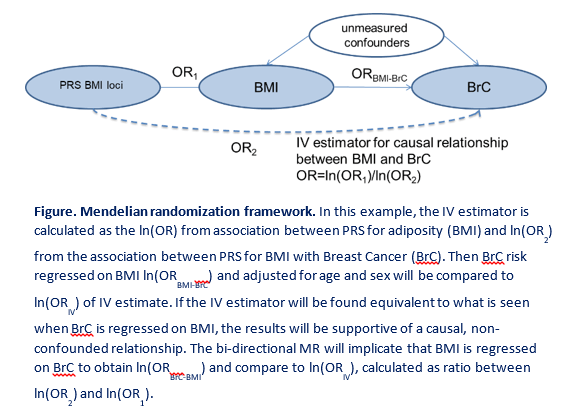
Growing observational and experimental evidence suggests that diabetes, obesity and cancers may share biological pathways in their pathogenesis. To disentangle the shared genetic component between diabetes and various types of cancer, we combine polygenic risk scores with hierarchical clustering of effects of these disease-associated DNA variants to disentangle the disease-specific genetic risk into its pathway-related components. We then test the effect of such pathway-related cumulative genetic risks on each disease and define the shared component. We also use Mendelian Randomisation (MR) to infer causal relationships between diabetes obesity, and cancers. Moreover, we implement multi-phenotype genome-wide association study (MPGWAS) approaches to yield a large improvement in the study power and help detecting shared genetic loci between co-morbid outcomes. Similarly, we study the relationships between the blood metabolite levels, diabetes and cancers. Additionally, we dissect the effects of each associated locus on all studied phenotypes to understand, whether the effect on multiple phenotypes, like that of TCF7L2 on diabetes and multiple cancers, is truly multi-phenotype or is mediated through a primary effect on one of the phenotypes. This is an exciting set of analytical efforts, which addresses a range important public health issues of disease comorbidity for better clinical care and prediction of the risks for comorbid conditions.
Within PreciDIAB, my research has already led to the publication of 4 papers, notably one in Nature Communication 4,5,6,7